Wholesale Bath and Body Supplies | Bulk Savings & Quality
Wholesale Bath and Body Supplies: Inside the Fizzy Boom
If you’re sourcing wholesale bath and body supplies, you’ve probably noticed the category’s odd mix of science and sparkle. Clean-beauty claims are up, novelty is still a thing (surprise toys, anyone?), and retailers want small MOQs with private label speed. I’ve toured a few factories; the good ones read like food-grade kitchens, not perfumeries.

Product spotlight: Safe Colorful Fizzies Organic Kids Children Bath Bombs
Origin: No. 18 Liuming Street, Xuefu Road, Chang’an District, Shi Jiazhuang, Hebei, China. Branded for OEM/ODM with private-label support. Officially tagged for the Adults age group, but formulated mild enough that many customers say their families use them together (always patch test, of course).

| Parameter | Specification (≈ / real-world use may vary) |
|---|---|
| Single package size | 6 × 6 × 6 cm |
| Single gross weight | ≈ 0.100 kg |
| Base materials | Sodium bicarbonate, citric acid, Epsom salt, organic shea butter, plant oils, food-grade colorants, optional natural fragrance |
| Function | Exfoliating, skin-softening, SPA relax |
| Customization | Shape, color, scent, surprise insert, logo, packaging |
| MOQ | Accepts small orders (pilot runs possible) |
| Shelf life | ≈ 18–24 months if kept sealed, cool, and dry |

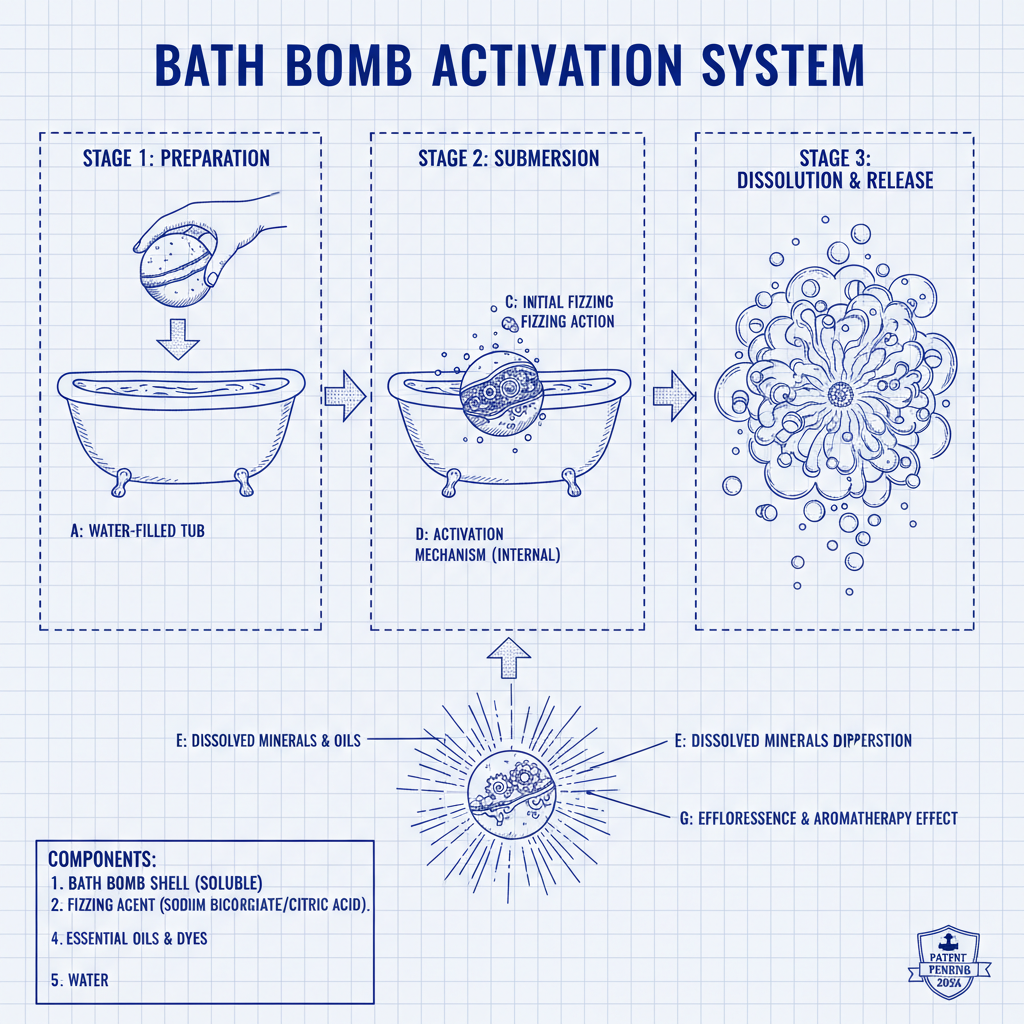
Process flow (what buyers actually ask about)
Materials: audited suppliers for citric acid and bicarbonate; plant oils and organic shea butter with batch COAs. Methods: dry blend → controlled hydration → press molding (custom dies) → low-humidity curing → wrap/seal. Surprise inserts are tested for leachables and sharp edges, then encapsulated. Testing: microbiological counts (ISO 21149/16212), preservative efficacy (ISO 11930, when applicable), HRIPT/patch testing by accredited labs, heavy metal screening per EU/US guidance, pH target ≈ 5.5–6.5. Service life validated via accelerated aging at 40°C/75% RH.
Compliance notes: ISO 22716 (cosmetic GMP) is the baseline; for U.S. retail, MoCRA facility registration and product listing apply; EU buyers expect CPSR and PIF under Regulation (EC) No 1223/2009. If there’s a toy-like surprise, suppliers often reference EN 71 parts 1–3. I’ve seen smart buyers ask for IFRA certificates for fragrances and allergen declarations—worth it.

Real-world test data (sample lot)
- Microbial counts:
- pH in solution: 5.8 (≈ room temp, 200 mL water)
- Colorant migration: none visible; bath staining rated 0/5 after rinse
- Dissolution time: around 3–5 minutes, tub temp 37–40°C

Vendor comparison (quick glance)
| Vendor | MOQ | Lead time | Compliance | Typical EXW | Notes |
|---|---|---|---|---|---|
| ENYU OEM (Hebei) | Low (accepts small orders) | ≈ 15–25 days after artwork | ISO 22716, MoCRA/EU doc support | $0.45–$0.95/bomb | Strong private label toolkit |
| Generic importer | Medium | ≈ 30–45 days | Basic COAs | $0.35–$0.70 | Cheaper, limited customization |
| Artisan microbrand | Very low | ≈ 7–14 days small runs | Local GMP practices | $1.20–$2.50 | High storytelling, higher cost |
Prices are indicative; real-world use may vary by formulation, packaging, and FX.

Use cases and quick wins
- Retail chains: seasonal gift boxes with custom scents and colors
- Hotels/spas: co-branded amenity kits; guests love the “surprise inside” angle
- Subscription boxes: limited drops; fast artwork swaps keep CAC down
Case study 1: a boutique hotel group switched to ENYU’s OEM pack in kraft tubes; repeat-purchase rate in lobby shops rose ≈ 19% month-on-month. Case study 2: a U.S. subscription brand moved from generic imports to wholesale bath and body supplies with IFRA-aligned fragrances; churn dipped a few points—small but profitable.
Why it sells (and keeps selling)
Fast customization, friendly MOQs, and consistent fizz. Honestly, that’s the trifecta. Buyers tell me dependable curing and clean rinse matter more than “viral glitter.” For wholesale bath and body supplies, documentation wins the PO: GMP, safety assessments, and clear labeling.
Citations
- ISO 22716: Cosmetics — Good Manufacturing Practices (GMP).
- FDA, Modernization of Cosmetics Regulation Act (MoCRA), facility registration & product listing guidance.
- ISO 11930: Evaluation of the antimicrobial protection of a cosmetic product.
- Regulation (EC) No 1223/2009 on cosmetic products; EU Cosmetic Product Safety Report (CPSR) requirements.